NGS Controls & Reference Materials
PGT-A
SHOP PRODUCTSPreimplantation genetic testing (PGT) or preimplantation genetic screening (PGS) both refer to the same process of testing embryos prior to their transfer into a patient during an in vitro fertilization (IVF) cycle. The aim of the method is to screen embryos for genetic anomalies and improve the odds of a successful pregnancy. There are three types of PGT testing that can be performed either individually or in combination:
- PGT-A - testing for chromosomal aneuploidies
- PGT-SR - testing for chromosomal structural rearrangements
- PGT-M - testing for monogenic disorders
Material obtained for PGT is small and very precious. It has to deliver accurate results to guide the decisions on embryo transfer. Most common way to obtain testing material is an embryo biopsy, done most commonly at the blastocyst stage (day 5). An alternative method relies on a non-invasive PGT by sampling embryo culture media (niPGT-A). Due to a small amount of genetic material obtained in the process, the first step of PGT is whole genome amplification (WGA).
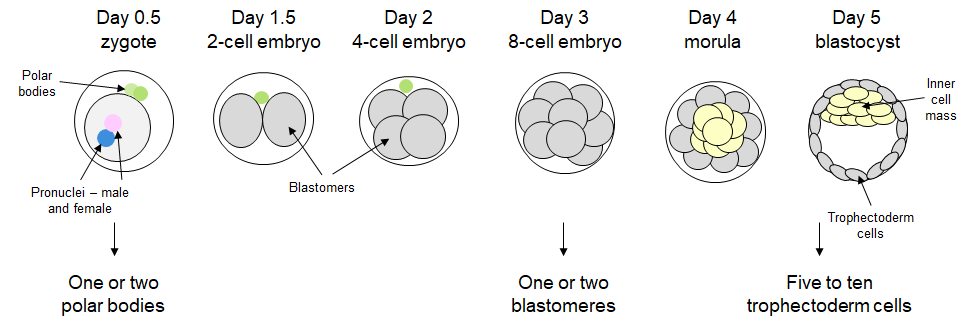
Figure 1. Early embryonic development with three most stages used for embryo biopsies.
LGC SeraCare now offers PGT-A reference materials for most common trisomies: 21, 18, 13 and euploid material which can be used as a negative control. These materials are:
- Formulated as genomic DNA from trisomic cells in buffer
- Offered at concentration suitable for the application
- Suitable for an introduction to PGT-A workflow at WGA step
- Compatible with an NGS-based PGT-A assays
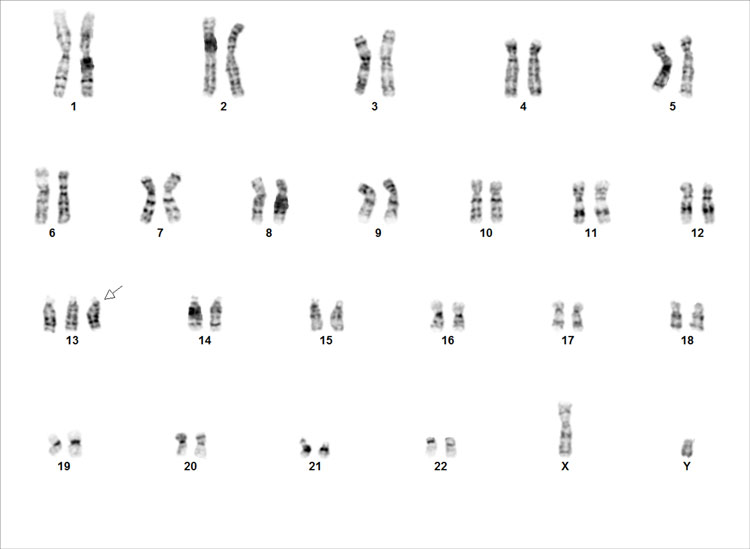
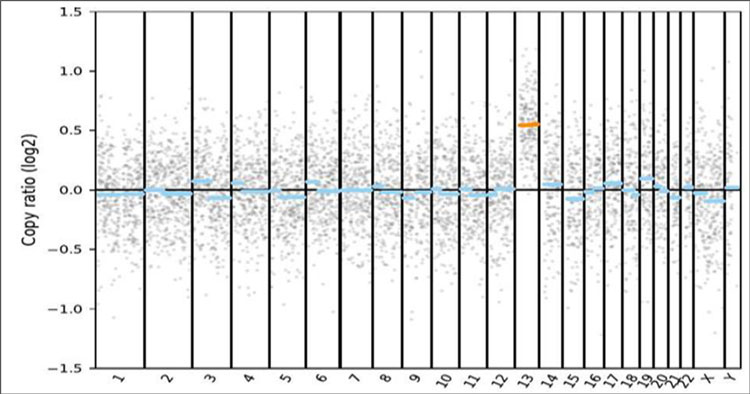
Figure 2A. Karyotype of the source cells. 2B. Performance of the gDNA material in a NGS-based PGT-A assay.
- Detect assay drift before results are affected with these specially formulated reference materials
- Accelerate assay implementation and troubleshooting with a sustainable source of highly consistent reference materials
- Reduce costs with easily customizable standards
- Use materials capable of benchmarking your test performance
