Somatic Cancer
Seraseq® ctDNA Extraction Reference Material 50 ng/mL
Details
Resources
Specifications
Preparation of a liquid biopsy sample starts with a blood draw from a patient followed by extraction of cfDNA from plasma. The concentration of cfDNA in a blood draw can vary greatly from patient to patient, but reference materials are typically built to a single cfDNA concentration. Additionally, extraction kits do not provide robust feedback about root cause of extractions resulting in lower than expected yield which increases the challenge of decoupling extraction kit performance from pre-analytic sample handling.
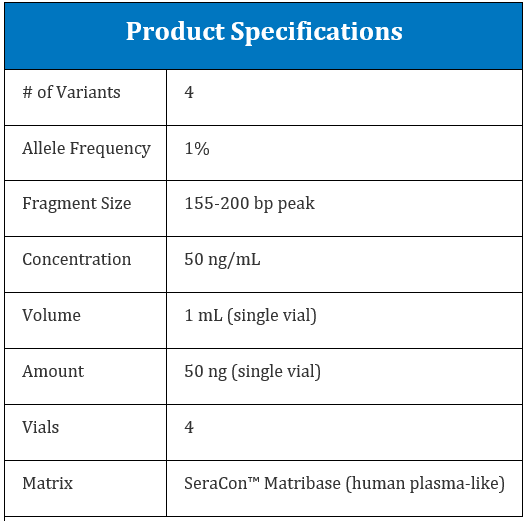
LGC Clinical Diagnostics has produced the Seraseq ctDNA Extraction Reference Materials to allow clinical labs to validate and assess the performance of cfDNA extractions in a more comprehensive manner. The ctDNA Extraction Reference Materials are available at concentrations of 20, 50 and 80 ng/mL in synthetic plasma.
In addition to being useful as total cfDNA extraction reference materials, the samples contain four EGFR variants present at 1% ctDNA to represent tumor ctDNA. Each kit comes with 4x single use vials for ease of use at an affordable price point.
Seraseq® ctDNA Extraction Reference Material 20 ng/mL, 50 ng/mL, 80 ng/mL
Seraseq® ctDNA Extraction Reference Material 50 ng/mL Product Sheet
Seraseq® ctDNA Extraction Reference Material 50 ng/mL
Seraseq ctDNA Extraction Reference Material Non-Requirement Letter
Technical Spreadhseet for Seraseq ctDNA Extraction Reference Material