Seroconversion Panels
AccuVert™ SARS-CoV-2 Seroconversion Panel
Details
Resources
If you already have an eCommerce account, please proceed with placing your order online. If you do not currently have an eCommerce account, please email your purchase order to CustomerService@Seracare.com or contact us at 1.800.676.1881.
AccuVert™ SARS-CoV-2 Seroconversion Panel is intended for use by diagnostic manufacturers, researchers, and clinical laboratories to develop, evaluate, or troubleshoot SARS-CoV-2 test methods. The panel consists of undiluted, naturally occurring plasma samples collected longitudinally from a single donor over the course of development and subsequent immunological response of a natural SARS-CoV-2 infection. Each sample represents a single collection event and has been evaluated extensively on leading commercial SARS-CoV-2 antibody tests.
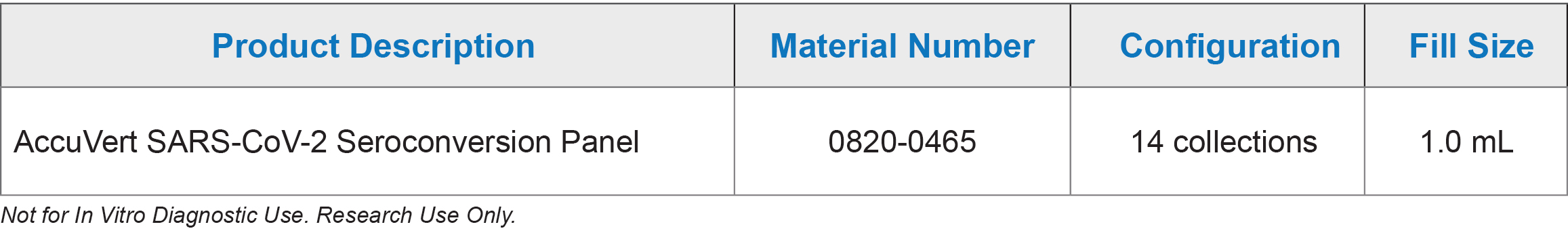
AccuVert SARS-CoV-2 Seroconversion Panel:
- Serially drawn, raw patient plasma specimens collected from a single donor during the development of a natural infection and subsequent response
- Detailed analyte evaluation with data-rich comparative test reports, enabling you to benchmark your assay against other industry-leading IVD kit developers



